 
The availability of suitable substitutes for a given commodity. A higher recycling rate may reduce risk to supply. The percentage of a commodity which is recycled. The number of atoms of the element per 1 million atoms of the Earth’s crust. This is calculated by combining the scores for crustal abundance, reserve distribution, production concentration, substitutability, recycling rate and political stability scores. 
The Chemical Abstracts Service registry number is a unique identifier of a particular chemical, designed to prevent confusion arising from different languages and naming systems.ĭata for this section been provided by the British Geological Survey.Īn integrated supply risk index from 1 (very low risk) to 10 (very high risk). Where more than one isotope exists, the value given is the abundance weighted average.Ītoms of the same element with different numbers of neutrons. This is approximately the sum of the number of protons and neutrons in the nucleus. The mass of an atom relative to that of carbon-12. The transition of a substance directly from the solid to the gas phase without passing through a liquid phase.ĭensity is the mass of a substance that would fill 1 cm 3 at room temperature. The temperature at which the liquid–gas phase change occurs. 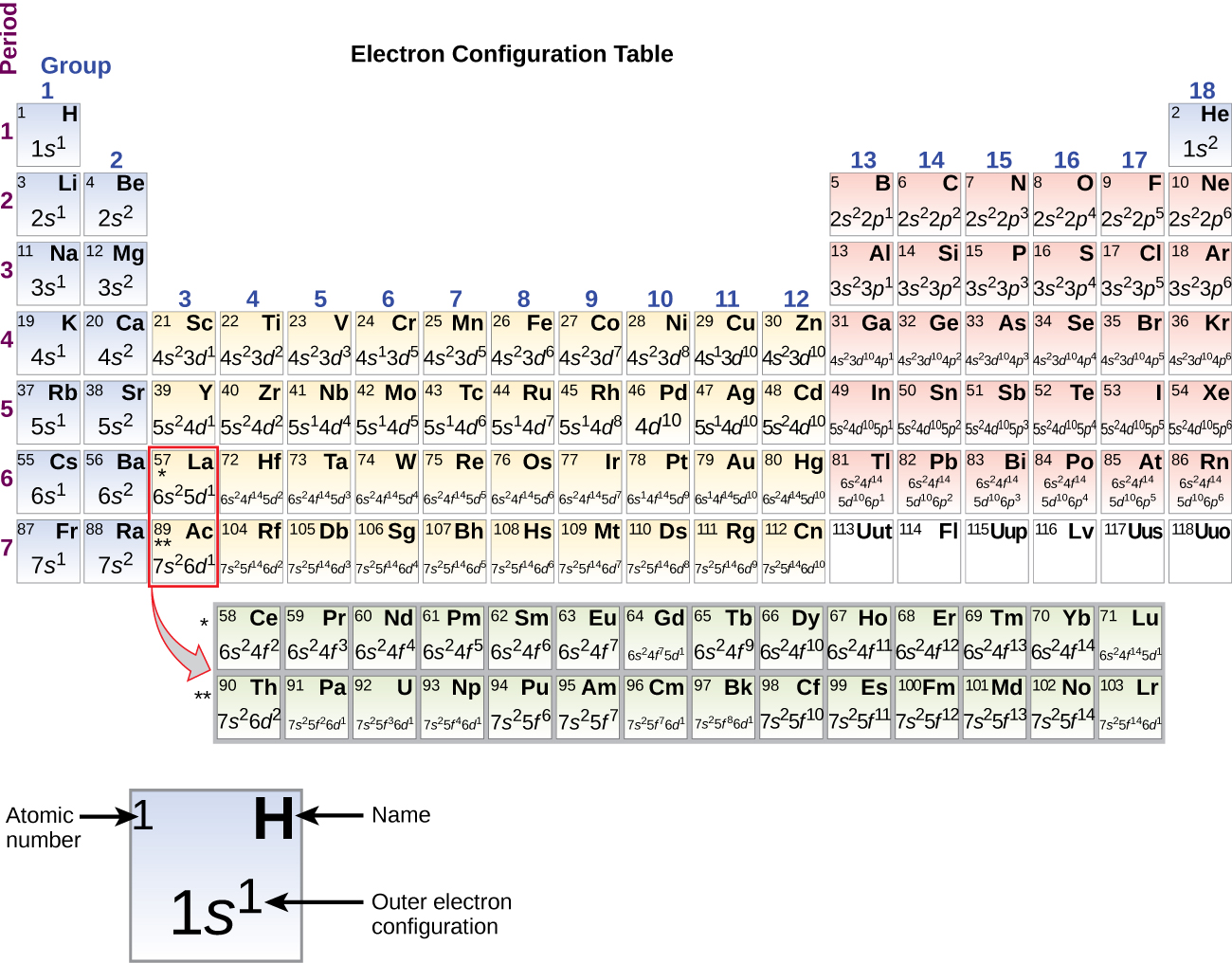
The temperature at which the solid–liquid phase change occurs. The arrangements of electrons above the last (closed shell) noble gas. These blocks are named for the characteristic spectra they produce: sharp (s), principal (p), diffuse (d), and fundamental (f). The atomic number of each element increases by one, reading from left to right.Įlements are organised into blocks by the orbital type in which the outer electrons are found. Members of a group typically have similar properties and electron configurations in their outer shell.Ī horizontal row in the periodic table. Working with elements from aluminum (which has an atomic number thirteen) to gold (seventy nine), he was able to show that the frequency of these transitions increased with each element studied.A vertical column in the periodic table. Moseley was able to confirm these two hypotheses through experimentation, measuring the wavelengths of photon transitions of various elements while they were inside an x-ray tube. The Bohr model of the atom had the central charge contained in its core, with its electrons circulating it in orbit, much like how the planet in the solar system orbit the sun. Two years later, Henry Moseley and Niels Bohr made further contributions that helped to confirm this. Antonius van den Broek added to this by formerly suggesting that the central charge and number of electrons were equal. This central charge would be roughly equal to half of the atoms total atomic weight. It was he who first suggested the model for an atom where the majority of its mass and positive charge was contained in a core. 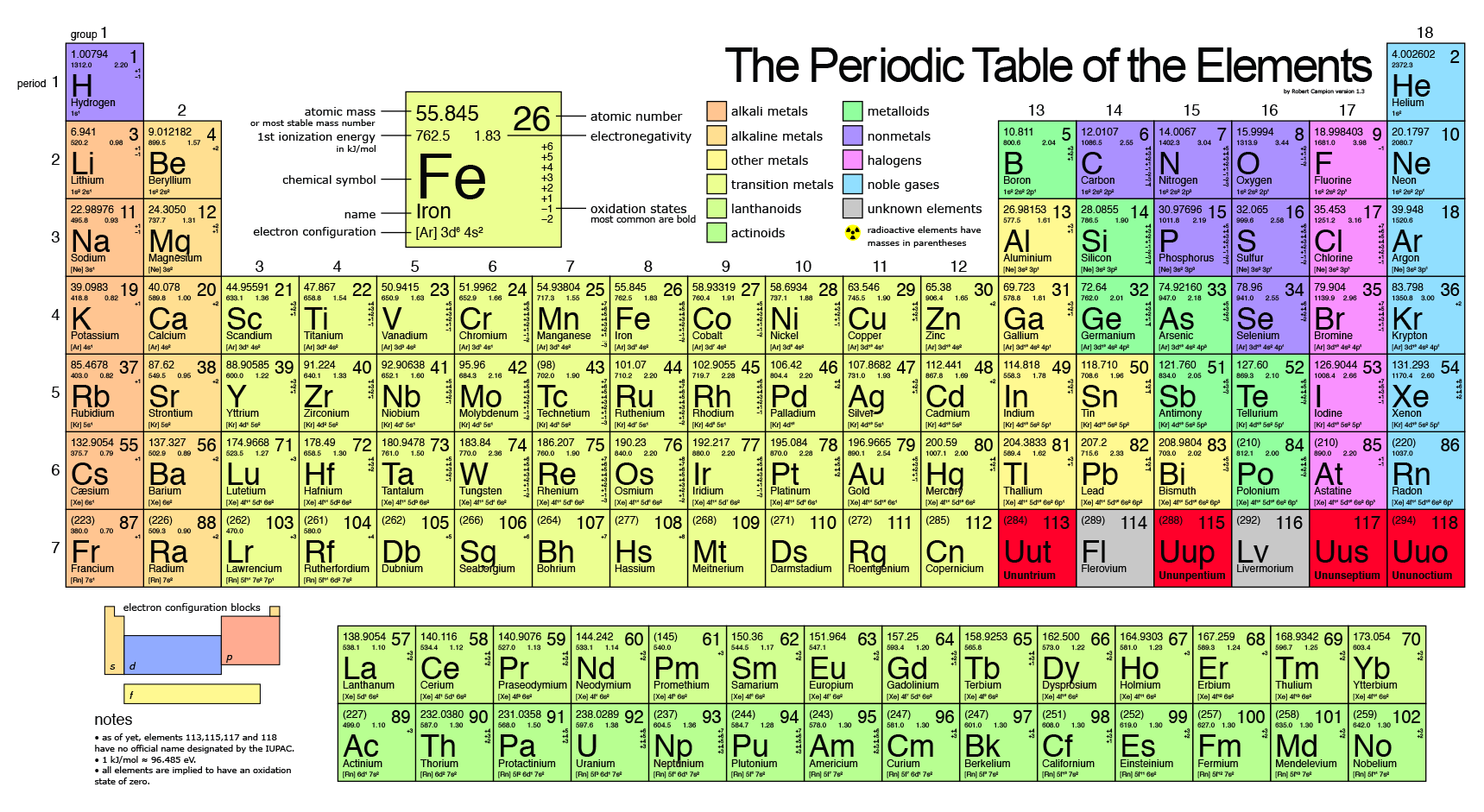
The atomic number of an element never changes, meaning that the number of protons in the nucleus of every atom in an element is always the same.Īrranging elements based on their atomic weight began with Ernest Rutherford in 1911. Oxygen atoms contain 8 protons and have an atomic number of 8, and so on. All carbon atoms contain six protons and therefore have an atomic number of 6. For example, Hydrogen atoms, which have one proton in their nucleuses, are given an atomic number of one. Ever wonder why the periodic table of elements is organized the way it is? Why, for example, does Hydrogen come first? And just what are these numbers that are used to sort them all? They are known as the element’s atomic number, and in the periodic table of elements, the atomic number of an element is the same as the number of protons contained within its nucleus. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
